https://doi.org/10.1140/epje/i2017-11565-9
Regular Article
The equation of state of polymers. Part III: Relation with the compensation law
Laboratoire de Physique des Solides, CNRS, Université de Paris-Sud, 91405, Orsay, France
* e-mail: jacques.rault@yahoo.fr
Received:
14
April
2017
Accepted:
21
August
2017
Published online:
29
September
2017
The properties of amorphous polymers and of organic compounds under pressure are interpreted in the framework of the modified Van der Walls Equation of State (mVW-EOS) the Vogel-Fulcher-Tamann (VFT) law and of the compensation law. We have shown recently that polymers and organic compounds in amorphous liquid and crystalline states verify the mVW-EOS which depends on three parameters, 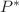
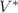 and
and  . In this paper we compare the characteristic pressure
. In this paper we compare the characteristic pressure 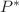 of the mVW-EOS to the various pressures
of the mVW-EOS to the various pressures  deduced from thermodynamic and kinetic properties of polymers in the liquid and solid states.
deduced from thermodynamic and kinetic properties of polymers in the liquid and solid states. 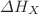 and
and 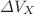 are: a) the enthalpy and volume change at the melting and glass transitions (the glass being isotropic or oriented and annealed below
are: a) the enthalpy and volume change at the melting and glass transitions (the glass being isotropic or oriented and annealed below 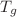 at various aging conditions); b) the activation parameters of individual
at various aging conditions); b) the activation parameters of individual 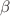 and cooperative
and cooperative 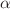 motions in crystalline liquid and amorphous polymers studied by dielectric or mechanical spectroscopy; and c) the activation parameters of amorphous (solid and liquid) polymers submitted to a deformation depending on the time frequency temperature and strain rate. For a same material, whatever its state and whatever the experimental properties analyzed (dielectric and mechanical relaxation, viscosity, auto-diffusion, yielding under hydrostatic pressure), we demonstrate that
motions in crystalline liquid and amorphous polymers studied by dielectric or mechanical spectroscopy; and c) the activation parameters of amorphous (solid and liquid) polymers submitted to a deformation depending on the time frequency temperature and strain rate. For a same material, whatever its state and whatever the experimental properties analyzed (dielectric and mechanical relaxation, viscosity, auto-diffusion, yielding under hydrostatic pressure), we demonstrate that  , (
, ( Grüneisen parameter,
Grüneisen parameter,  compressibility). In all polymers and organic compounds (and water), these pressures, weakly dependent on T and P near
compressibility). In all polymers and organic compounds (and water), these pressures, weakly dependent on T and P near 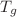 and
and 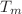 at low pressure are characteristic of the H-H inter-molecular interactions. It is shown that the two empirical Lawson and Keyes relations of the compensation law can be deduced from the mVW-EOS.
at low pressure are characteristic of the H-H inter-molecular interactions. It is shown that the two empirical Lawson and Keyes relations of the compensation law can be deduced from the mVW-EOS.
Key words: Soft Matter: Polymers and Polyelectrolytes
© EDP Sciences, SIF, Springer-Verlag GmbH Germany, 2017





